ABSTRACT
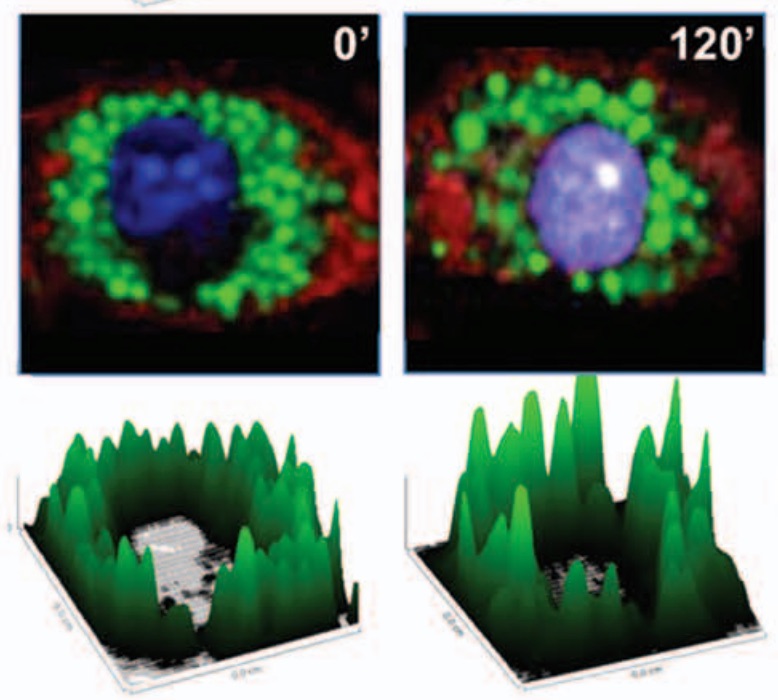
The Nod-like receptor, Nlrp3, has been linked to inflammatory diseases and adjuvant-mediated immune responses. A wide array of structurally diverse agents does not interact directly with Nlrp3, but is thought to activate the Nlrp3 inflammasome by inducing a common upstream signal, such as lysosome rupture. To test the connection between lysosome integrity and Nlrp3 signaling, we analyzed inflammasome activation following stimulation of murine macrophages with lysosome-destabilizing agents and pyroptosis inducers. Here we provide evidence that lysosomal rupture and the corresponding release of lysosomal hydrolases is an early event in macrophages exposed to the lysosome-destabilizing adjuvants LLOMe and alum. Lysosome rupture preceded cell death induction mediated by these agents and was associated with the degradation of low-molecular weight proteins, including the inflammasome component caspase-1. Proteolysis of caspase-1 was controlled by specific cathepsins, but was independent of autocatalytic processes and Nlrp3 signaling. Consistent with these findings, lysosome-disrupting agents triggered only minimal caspase-1 activation and failed to cause caspase-1-dependent cell death (pyroptosis), generally associated with Nlrp3 signaling. In contrast, lysosome rupture was a late event in macrophages exposed to prototypical pyroptosis inducers. These agents triggered extensive Nlrp3 signaling prior to lysosome rupture with only minimal impact on the cellular proteome. Taken together, our findings suggest that lysosome impairment triggers a cascade of events culminating in cell death but is not crucial for Nlrp3 signaling. The significant differences observed between lysosome-disrupting agents and pyroptosis inducers might explain the distinct immunologic responses associated with these compounds.